Click Here to Find a Vaccine Location
Please use the GA Dept of Public Health – Vaccine Locator via the link above. Search by county to find Vaccine locations convenient for you. Be sure to check back often as the COVID participating vaccine site locations list will be updated frequently. Additional locations statewide will be added when providers are ready to safely administer the vaccine, and as vaccine supply allows.
Why should I get a COVID Vaccine?
The COVID vaccines that are currently offered are 95% effective at reducing the likelihood of developing symptomatic COVID. During research studies, these vaccines also dramatically reduced the number of severe cases of COVID-19. Getting aCOVID vaccine will help protect you, your family, your friends, coworkers, and neighbors.
As of January 28, 2021, there have been 25,456,670 reported new infections and 427,626 covid related deaths in the United States.
(CLICK HERE TO VIEW REAL-TIME CDC COVID19 DATA)
COVID Vaccinations in Pregnancy – Click for more info
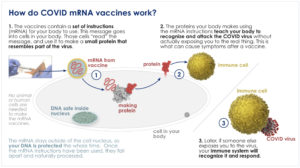
How do COVID mRNA vaccines work?
How were COVID mRNA vaccines tested?
Emory University was one of the earliest test sites for COVID mRNA vaccines. Diverse members of our community, medical professionals, and others around the world volunteered to be a part of the testing of the vaccines and aided with their development. Outside experts such as scientists at the CDC and FDA carefully monitored the process according to rigorous standards. Many of the people who helped develop and test the vaccine have also rolled up their sleeves to get it. All of their findings were made public and are available online for outside experts (and you) to examine. This has been and continues to be an open, visible process.
How did they develop these vaccines so quickly?
- University researchers had already been studying the science behind mRNA vaccines for decades.
- To make most vaccines, scientists have to grow the whole virus or produce parts of it, which takes time. This mRNA vaccine doesn’t use any part of the virus, so nothing needed to be grown.
- A lot of Americans volunteered to test the vaccines.
- Pfizer: 42% of global participants and 30% of U.S. participants have racially and ethnically diverse backgrounds; Moderna: 11,000 (37%) study participants were from communities of color.
- COVID was spreading very quickly in American communities, so it took less time than usual for people in the studies to be exposed to COVID to see if they had immunity.
- The US government took on the financial risk of producing vaccines ahead of time. This meant companies were willing to manufacture vaccines right away, instead of waiting for approvals.
What are the side effects and risks?
Hundreds of thousands of Americans have already had COVID vaccines before you, including almost 40,000 people in 2020 during the testing phase of the vaccines which we call clinical trials.
- It is NOT possible to get COVID from the vaccines.
- 85-90% of people who got the vaccines either did not report feeling any side effects or had only mild symptoms that didn’t interfere with daily activities.
- If you do experience side effects, it is generally a sign that your immune system is working well. They occur more often after the second dose. This may include discomfort at the injection site, some tiredness, mild headache, muscle or joint pain, swollen lymph nodes, fevers/chills, or a general feeling of being unwell. As with all vaccines, the risk of having an allergic reaction is very small.
- Pregnant women were not included in the initial studies, but there is no suggestion of particular risks. They can receive the vaccination and should discuss any questions with their doctor.

Click Here to Find a Vaccine Location
Please use the GA Dept of Public Health – Vaccine Locator via the link above. Search by county to find Vaccine locations convenient for you. Be sure to check back often as the COVID participating vaccine site locations list will be updated frequently. Additional locations statewide will be added when providers are ready to safely administer the vaccine, and as vaccine supply allows.
Myths and Facts about COVID-19 Vaccines
Can a COVID-19 vaccine make me sick with COVID-19
No. None of the authorized COVID-19 vaccines in the United States contain the live virus that causes COVID-19. This means that a COVID-19 vaccine cannot make you sick with COVID-19.
COVID-19 vaccines teach our immune systems how to recognize and fight the virus that causes COVID-19. Sometimes this process can cause symptoms, such as fever. These symptoms are normal and are signs that the body is building protection against the virus that causes COVID-19.
Will getting a COVID-19 vaccine cause me to test positive for COVID-19 on a viral test? 
No. None of the authorized and recommended COVID-19 vaccines cause you to test positive on viral tests, which are used to see if you have a current infection.
If your body develops an immune response to vaccination, which is the goal, you may test positive on some antibody tests. Antibody tests indicate you had a previous infection and that you may have some level of protection against the virus.
Is it safe for me to get a COVID-19 vaccine if I would like to have a baby one day?
Yes. If you are trying to become pregnant now or want to get pregnant in the future, you may get a COVID-19 vaccine when one is available to you.
There is currently no evidence that COVID-19 vaccination causes any problems with pregnancy, including the development of the placenta. In addition, there is no evidence that female or male fertility problems are a side effect of any vaccine, including COVID-19 vaccines.
Will a COVID-19 vaccine alter my DNA?
No. COVID-19 vaccines do not change or interact with your DNA in any way. Both mRNA and viral vector COVID-19 vaccines deliver instructions (genetic material) to our cells to start building protection against the virus that causes COVID-19. However, the material never enters the nucleus of the cell, which is where our DNA is kept.
Do any of the COVID-19 vaccines authorized for use in the United States shed or release any of their components?
No. Vaccine shedding is the term used to describe the release or discharge of any of the vaccine components in or outside of the body. Vaccine shedding can only occur when a vaccine contains a weakened version of the virus. None of the vaccines authorized for use in the U.S. contain a live virus. mRNA and viral vector vaccines are the two types of currently authorized COVID-19 vaccines available.
Can receiving a COVID-19 vaccine cause you to be magnetic?
No. Receiving a COVID-19 vaccine will not make you magnetic, including at the site of vaccination which is usually your arm. COVID-19 vaccines do not contain ingredients that can produce an electromagnetic field at the site of your injection. All COVID-19 vaccines are free from metals.
Learn more about the ingredients in the COVID-19 vaccinations authorized for use in the United States.
Last Updated July 7, 2021
Content source: National Center for Immunization and Respiratory Diseases (NCIRD), Division of Viral Diseases